 |
 |
- Search
| Precis Future Med > Volume 6(4); 2022 > Article |
|
Abstract
Monogenic diabetes is commonly caused by single-gene mutations. This disease ranges from 1% to 5% in all cases of diabetes and is less affected by behavior and environment. Neonatal diabetes mellitus (NDM) and maturity-onset diabetes of the young (MODY) account for a major proportion of monogenic diabetes, while syndromic diabetes constitutes a smaller proportion. Diagnosis of monogenic diabetes has improved from being based on clinical phenotypes to molecular genetics, with significant advancement of genome sequencing skills. Precise medication for monogenic diabetes is based on genetic testing; therefore, an accurate diagnosis is essential. Due to the basic clinical criteria (diagnosed < 6 months of age), genetic testing and precision treatment for NDM are fast and uncomplicated. The MODY probability calculator was developed; however, it remains challenging to distinguish MODY from type 1 and 2 diabetes due to the lack of a single diagnostic criteria and genetic testing. Additionally, the high cost and complicated interpretation of these genetic test results add to these challenges. This review will discuss the distinct etiology and subgroups that contribute to predicting and treating clinical phenotypes associated with monogenic diabetes. Furthermore, we will review the recent Korean studies and suggest methods of prioritizing patient screening for genetic testing.
Monogenic diabetes is commonly caused by single-gene mutations with more than 50 causative genes identified. This disease ranges from 1% to 5% in all cases of diabetes and is less affected by behavior and environment [1,2]. Neonatal diabetes mellitus (NDM) and maturity-onset diabetes of the young (MODY) account for a major proportion of monogenic diabetes, while syndromic diabetes constitutes a smaller proportion [3,4]. Over the past decades, diagnosis of monogenic diabetes has improved from being based on clinical phenotypes to molecular genetics, with significant advancement of genome sequencing skills.
Precision medicine is based on tailoring treatment for evidence-based individualization [5]. Challenges faced when implementing precision medicine in diabetes are disease heterogeneity, various genetic influences, difficulties of accurate diagnosis, limitations of current treatment, and social, environmental, and psychological factors [6]. This review will explain how successfully precision medicine is implemented in monogenic diabetes by examining the distinct etiology and subgroups that contribute to predicting and treating clinical phenotypes associated with the disease. Furthermore, we will also review the recent Korean studies and suggest methods of prioritizing patient screening for genetic testing.
NDM is defined as developing within the first 6 months of life. Before the availability of genetic screening, NDM was classified based on the clinical course of transient NDM (TNDM), permanent NDM (PNDM), or a specific syndrome. The development of targeted sequencing has allowed for rapid diagnosis of all known genetic etiologies and subsequent precision treatment for NDM [7-10].
Patients with TNDM by 6q24 methylation are characterized by hyperglycemia during the first week of life, which resolves by age 18 months [7]. Many of them require exogenous insulin treatment at diagnosis and during remission, while some of them are well responsive to sulfonylurea or a combination of sulfonylurea and inulin. However, approximately 50% of patients relapse during late childhood or early adulthood; therefore, an accurate record of the patient’s history of TNDM is essential. There is no guideline for treatment for relapse, some patients can be treated with sulfonylurea or a combination of sulfonylurea and insulin [11,12]. Commonly, TNDM is caused by overexpression of imprinted genes on chromosome 6q24 (pleomorphic adenoma gene 1 [PLAG1] and hydatidiform mole associated and imprinted [HYMAI]) due to paternally inherited duplications, paternal uniparental disomy, or maternal hypomethylation. PLAG1 and HYMAI encode a zinc finger protein (ZEP) and long noncoding RNA, respectively. Notably, patients with mutations on 6q24 have low birthweight and early development compared to patients with potassium adenosine triphosphate (KATP) channel mutations [13]. The potassium inwardly rectifying channel subfamily J member 11 (KCNJ11) gene encoding the Kir6.2 subunits of the KATP channel and ATP binding cassette subfamily C member 8 (ABCC8) encoding the sulfonylurea receptor 1 (SUR1) subunit of the KATP channel, whose mutations induce PNDM more frequently, are the remaining causes of TNDM.
The most frequent causes of PNDM are heterozygous gain-of function mutations in KCNJ11 and ABCC8, while homozygous or some heterozygous loss-of-function mutations in KCNJ11 and ABCC8 develop congenital hyperinsulinism [9,14]. These KATP mutations impede KATP channel closure in multiple ways in both TNDM and PNDM [15]. Additionally, mutations in ABCC8 are more prevalent in TNDM, while mutations in KCNJ11 are more prevalent in PNDM. In TNDM and PNDM, KATP-related diabetes responds well to high-dose sulfonylureas rather than insulin.
PNDM is associated with specific syndromes. Immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome is caused by forkhead box P3 (FOXP3), which is an autoimmune disease characterized by persistent diarrhea, hemolytic anemia, exfoliative dermatitis, autoimmune thyroiditis, and PNDM. Wolcott-Rallison syndrome is an inherited autosomal recessive disease caused by a mutation of eukaryotic translation initiation factor 2-alpha kinase 3 (EIF2AK3), which leads to pancreatic β-cell apoptosis, as well as other clinical characteristics, including mental retardation, renal failure, and early mortality [16]. Approximately 80% of patients with PNDM have known genetic etiology; hence, whole exome sequencing remains a continuous effort [17].
MODY is the most prevalent form of monogenic diabetes and was first identified in 1974 by Fajans and Bell [3] and Tattersall [18]. The prevalence of MODY is estimated to be at least 1.2% according to a population-based study conducted in the United States [1]. Currently, 14 genes are identified as disease-causing mutations in MODY patients (Table 1) [19,20]. According to the most comprehensive study that was conducted in the United Kingdom, hepatocyte nuclear factor 1 α (HNF1A, 52%) and glucokinase (GCK, 32%) are the most common causes of MODY, followed by HNF4α (10%) and HNF1β (6%) [21]. It is thought that the study methodology, such as body mass index (BMI), cut-off age, and genetic testing method, influence the prevalence and subtype of MODY between ethnic groups and conducted studies.
GCK-MODY is caused by heterozygous inactivating mutations in the glucose sensor enzyme GCK [22]. GCK stimulates glucose metabolism and promotes insulin secretion through multiple mechanisms, including protein stability, enzymatic activity, and increased interaction with the GCK regulator. Less heterogeneity is observed in patients with GCK mutations, who have mild, lifelong, asymptomatic fasting hyperglycemia in the pre-diabetes range. Some patients exhibit diabetes due to type 2 mixed with environmental and hereditary factors. Hyperglycemia in GCK-MODY is less responsive to insulin and oral agents and does not appear to be associated with micro- or macrovascular complications; therefore, these patients seldom need glucose-lowering treatment, except during pregnancy [23]. Maternal GCK mutations can result in macrosomia due to increased insulin secretion in response to hyperglycemic uterine conditions. However, insulin treatment during pregnancy increases the risk of low gestational age in a fetus with GCK mutations; due to the decreased insulin secretion and glucose level below the fetal set point [24]. Therefore, it is ideal to know whether a fetus has mutations. Recently, droplet digital Polymerase chain reaction (PCR) has been used to determine fetal mutations noninvasively by extracting cell-free DNA from a maternal blood sample [25].
HNF1A-MODY encodes homeodomain-containing transcription factors related to β-cell development and insulin secretion and is the most prevalent form of monogenic diabetes in the United Kingdom [21]. Over 400 variants of HNF1A have been identified [26]. HNF1A is commonly diagnosed at an earlier age and with a lower BMI than type 2 diabetes, although the risk of long-term micro- and macrovascular complications is similar [27]. HNF1A regulates the expression of sodium-glucose cotransporter 2 (SGLT2), resulting in glycosuria without definite hyperglycemia [28]. Therefore, early glycosuria can serve as a diagnostic indicator for patients with HNF1A mutations, and caution is required when treating these patients with SGLT2 inhibitors [29].
HNF4A-MODY is caused by mutations in genes that encode hepatocyte nuclear factor 4α. These variants are less common than HNF1A mutations but have similar clinical features and treatments. Patients with HNF1A or HNF4A variants have an excellent response to sulfonylurea, which binds to the potassium channel and depolarize the β-cell, resulting in insulin secretion [30]. The standard sulfonylurea dose used to treat type 2 diabetes can cause hypoglycemia in these patients; therefore, a lower dose is recommended. Despite sharing similar characteristics, neonates with HNF4A mutations show increased insulin secretion, leading to macrosomia and transitory hyperinsulinemic hypoglycemia. However, after neonatal hyperinsulinemia occurs, insulin production decreases, and diabetes develops; this cause of diabetes remains unclear [31]. A glucagon-like peptide 1 receptor agonist or dipeptidyl peptidase-4 inhibitor with sulfonylurea was effective without occurring hypoglycemia in HNF1A-MODY, suggesting that HNF1A or HNF4A-MODY patients may benefit from treatment [32,33].
HNF1B mutations are highly uncommon, accounting for fewer than 1% of MODY patients. HNF1B variants cause MODY type 5 and are associated with multiorgan disease, including renal abnormalities (predominantly renal cysts), abnormal liver function tests, and neurocognitive defects. Approximately 50% of these variants are due to mutations in the HNF1B gene, while the remainder is due to a 17q12 deletion spanning 15 genes, including HNF1B [34]. Although some patients initially respond to sulfonylureas or repaglinide, pancreatic hypoplasia and hepatic insulin resistance eventually necessitate insulin therapy [35]. Therefore, not only diabetic specialists but also nephrologists, urologists, and gynecologists should manage HNF1B-MODY.
Monogenic diabetes can develop multisystem syndromes. Diabetes can be a first manifestation of multisystem syndromes.
Wolfram syndrome is a rare disease caused by recessive mutations in the wolframin ER transmembrane glycoprotein (WFS1) [36]. This syndrome is characterized by diabetes insipidus, optic atrophy, insulin-dependent diabetes, and sensorineural deafness. Diabetes is usually diagnosed in the first decade of life, and accurate and prompt diagnosis is necessary due to the high morbidity and mortality rates. Recently, some WFS1 mutations have been reported to cause isolated diabetes with reduced penetrance for other wolfram syndrome related features [37].
Mitochondrial diabetes is also known as maternally inherited diabetes and deafness. This type of diabetes is caused by a pathogenic mutation in the mitochondrial DNA, usually m.3243A>G [38]. Diabetes is typically diagnosed in the third or fourth decades of life. The hearing loss is usually diagnosed before diabetes, and is bilateral, sensorineural, and progressive [39]. Other rare forms of mitochondrial diabetes comprise of cardiomyopathy, myopathy, nephropathy, and central neurological features in a rare form of mitochondrial diabetes. The penetrance is estimated to be approximately 100% by the age of 70 years. These patients have impaired insulin secretion; therefore, insulin treatment is necessary for most patients.
Monogenic lipodystrophy is a heterogeneous group, characterized by a complete or partial lack of adipose tissue and adipose tissue-derived hormones. This develops insulin resistance and metabolic complications. Due to the lack of adipose tissue, dyslipidemia results in insulin resistance by spillover of fat into ectopic areas [40]. These patients can be classified into congenital generalized lipodystrophy (CGL) and familial partial lipodystrophy (FPLD). CGL is an autosomal recessive disorder and is usually caused by mutations in 1-acylglycerol-3-phosphate O-acyltransferase 2 and Berardinelli-Seip congenital lipodystrophy 2 [41]. These patients have common clinical features such as diabetes, generalized lipodystrophy, and dyslipidemia. On the other hand, FPLD is typically caused by lamin A/C or peroxisome proliferator-activated receptor γ. These patients have body fat deficiency on the buttocks, hips, and limbs.
The diagnosis of monogenic diabetes has increased but is still gradual, although genetic testing is quite common even during pregnancy [25,42]. Based on genetic testing, precise treatment for monogenic diabetes has become a novel concept for diabetologists. The International Society for Pediatric and Adolescent Diabetes recommends genetic testing for diabetes patients younger than 6 months and without autoantibodies if younger than 12 months [43]. Moreover, it helps the clinician predict prognosis and improve care, including for patients with KATP mutations treated with oral sulfonylurea [10]. The genetic test is cost-effective for these young patients. However, evaluating young adults and children with type 1 and type 2 diabetes presents certain difficulties, although onset age, endogenous insulin secretion, absence of obesity, and absence of autoantibodies help distinguish MODY from type 1 and type 2 diabetes [21,44,45]. Consequently, there is no single criteria that may accurately identify MODY patients. Using the ideal cut-offs, Shield et al. [46] developed the ‘MODY Probability Calculator,’ which may be accessed for free at www.Diabetesgenes.org. Additionally, incorporating MODY characteristics into this model can result in an improved diagnosis of MODY.
Using a next-generation sequencing (NGS) panel, a patient’s entire exome or all MODY genes can be examined simultaneously. Sanger confirmation may be required after variants are found in NGS; however, exome analysis can miss mutations in noncoding regions, deep intronic regions, and 5’- and 3’-untranslated regions (UTRs) associated with monogenic diabetes [47]. Using exome or genome sequencing for monogenic diabetes frequently identifies novel variants; therefore, caution must be exercised when differentiating disease-causing mutations from benign variants. The American College of Medical Genetics and Genomics and the Association for Molecular Pathology published guidelines for interpreting sequence variants in 2015 [48]. According to these guidelines, variants must be classified as pathogenic, likely pathogenic, of uncertain significance, likely benign, and benign. After the first publication of guidelines, revised recommendations regarding loss-of-function and uses of functional evidence were provided [49,50].
High-sensitivity C-reactive protein (hs-CRP) is emerging as a novel biomarker for MODY [51]. Moreover, an HNF1α binding site in the CRP gene is involved in regulating the basal constitutive CRP in the liver [52]. Patients with HNF1A variants have significantly lower hs-CRP than others, including type 1 diabetes, type 2 diabetes, and GCK-MODY [53,54]. However, the Singapore study did not have a better diagnostic yield based on the hs-CRP criteria. It is assumed that various factors, such as BMI and race, affect the hs-CRP level [55]. Therefore, the appropriate cut-off value for hs-CRP is needed, and caution is required for its efficacy in the Asian population in particular. Additionally, plasma fucosylated glycan and apolipoprotein M are promising candidates [56,57]. Recently, microRNA (miRNAs) has been used as a promising biomarker; Bonner et al. [58] reported the use of circulating miRNAs in a patient with HNF1A-MODY. Furthermore, miRNA has been reported as a gene regulator in diabetes [59].
The most important key element of diagnosing monogenic diabetes is identifying disease-causing genes that can classify patients into particular subgroups [60]. Different treatments can significantly affect the outcomes in these subgroups due to the efficacy of precision medicine in these subgroups with known etiology. Additionally, accurate diagnosis improves clinical prognosis and family genetic counseling. The phenotype of KCNJ11 neonatal diabetes depends on the severity of the mutation [61]. HNF4A-MODY patients with the p.R114W mutation have different phenotypes, showing decreased sensitivity to low-dose sulfonylurea, lower penetrance, and no effect on birthweight when compared to patients with other HNF4A-MODY mutations [62].
In Korea, the first genetic study for MODY was conducted in 2001 and focused on HNF1α [63]. Thereafter, various attempts were made to diagnose MODY patients [64,65]. Since 2015, whole exome sequencing has been utilized to identify candidate genes for MODY [66-68]. Recent studies using a targeted gene panel have been undertaken on Korean patients suspected of having monogenic diabetes [69]. This study reported that among 109 patients with probable monogenic diabetes, 23 (21.1%) carried pathogenically or possibly pathogenically mutations, with GCK-MODY being the most prevalent (50%). These results are similar to those of a comprehensive study on monogenic diabetes conducted in the United Kingdom [21]. According to a recent report from Japan, 39.2% were diagnosed with MODY; however, 18.4% showed de novo mutations contrary to the standard MODY criteria in this study [70]. Although standard MODY criteria include early onset (before age 25 to 35), absence of insulin dependence, absence of obesity and autoantibodies, and autosomal dominant inheritance for at least two generations [71], approximately 1% of MODY patients have glutamate decarboxylase antibodies, de novo mutations, and obesity, which make it challenging for the clinician to diagnose monogenic diabetes [72]. Approximately 60% to 80% of MODY’X’ was not identified in suspected monogenic diabetes patients, there is continuous attempt to find out unidentified genes for MODY. The precision diabetes treatment approach may still be challenging, and until genetic testing can be performed on every suspected patient with monogenic diabetes, the physician should prioritize patients with high suspicion through their biomarkers, probability score, or clinical characteristics. Regarding these difficulties, a proposed diagnostic algorithm for genetic testing in patients with diabetes could be helpful to the clinical community (Fig. 1).
Notes
AUTHOR CONTRIBUTIONS
Conception or design: KMJ.
Acquisition, analysis, or interpretation of data: KMJ.
Drafting the work or revising: KMJ.
Final approval of the manuscript: KMJ.
Fig. 1.
Suggested diagnostic algorithm for monogenic diabetes for genetic testing. NDM, neonatal diabetes mellitus; MODY, maturity-onset diabetes of young; GCK, glucokinase.
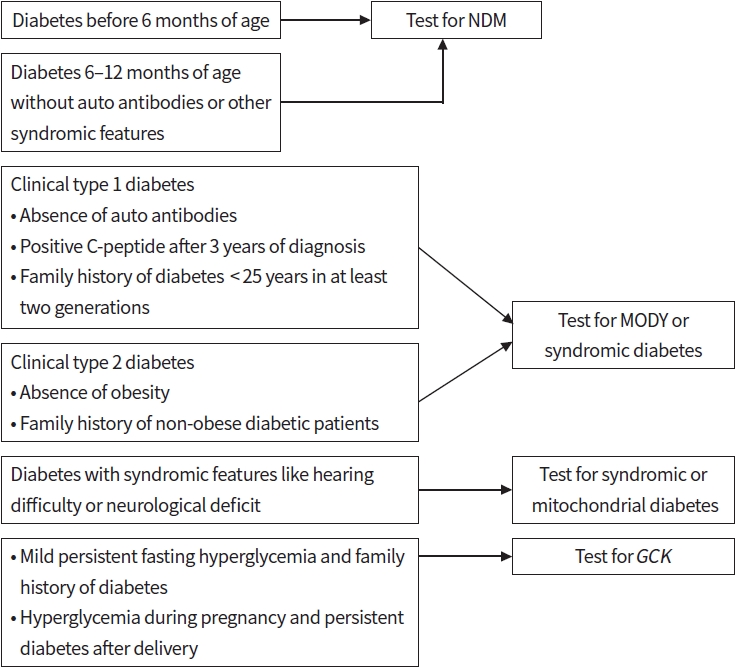
Table 1.
Each gene and subtype of maturity-onset diabetes of the young and clinical characteristics
Adapted from Urakami [19].
MODY, maturity-onset diabetes of young; OAD, oral antidiabetic drug; PNDM, permanent neonatal diabetes mellitus; ATP, adenosine triphosphate.
REFERENCES
1. Pihoker C, Gilliam LK, Ellard S, Dabelea D, Davis C, Dolan LM, et al. Prevalence, characteristics and clinical diagnosis of maturity onset diabetes of the young due to mutations in HNF1A, HNF4A, and glucokinase: results from the SEARCH for Diabetes in Youth. J Clin Endocrinol Metab 2013;98:4055–62.



2. Kanakatti Shankar R, Pihoker C, Dolan LM, Standiford D, Badaru A, Dabelea D, et al. Permanent neonatal diabetes mellitus: prevalence and genetic diagnosis in the SEARCH for Diabetes in Youth Study. Pediatr Diabetes 2013;14:174–80.

3. Fajans SS, Bell GI. MODY: history, genetics, pathophysiology, and clinical decision making. Diabetes Care 2011;34:1878–84.


4. Suh J, Choi HS, Kwon A, Chae HW, Lee JS, Kim HS. A novel compound heterozygous mutation of the AIRE gene in a patient with autoimmune polyendocrine syndrome type 1. Ann Pediatr Endocrinol Metab 2019;24:248–52.




5. National Research Council (US) Committee on A Framework for Developing a New Taxonomy of Disease. Toward precision medicine: building a knowledge network for biomedical research and a new taxonomy of disease. Washington (DC): National Academies Press; 2011.
7. Polak M, Cave H. Neonatal diabetes mellitus: a disease linked to multiple mechanisms. Orphanet J Rare Dis 2007;2:12.




8. Zhang H, Colclough K, Gloyn AL, Pollin TI. Monogenic diabetes: a gateway to precision medicine in diabetes. J Clin Invest 2021;131:e142244.



9. Mouron-Hryciuk J, Stoppa-Vaucher S, Busiah K, Bouthors T, Antoniou MC, Jacot E, et al. Congenital hyperinsulinism: 2 case reports with different rare variants in ABCC8. Ann Pediatr Endocrinol Metab 2021;26:60–5.




10. Abdelmeguid Y, Mowafy EW, Marzouk I, Franco E, ElSayed S. Clinical and molecular characteristics of infantile-onset diabetes mellitus in Egypt. Ann Pediatr Endocrinol Metab 2022;27:214–22.




11. Fu JL, Wang T, Xiao XH. Relapsed 6q24-related transient neonatal diabetes mellitus successfully treated with sulfonylurea. Chin Med J (Engl) 2019;132:846–8.



12. Carmody D, Beca FA, Bell CD, Hwang JL, Dickens JT, Devine NA, et al. Role of noninsulin therapies alone or in combination in chromosome 6q24-related transient neonatal diabetes: sulfonylurea improves but does not always normalize insulin secretion. Diabetes Care 2015;38:e86–7.




13. Docherty LE, Kabwama S, Lehmann A, Hawke E, Harrison L, Flanagan SE, et al. Clinical presentation of 6q24 transient neonatal diabetes mellitus (6q24 TNDM) and genotype-phenotype correlation in an international cohort of patients. Diabetologia 2013;56:758–62.



14. Mooij CF, Tacke CE, van Albada ME, Barthlen W, Bikker H, Mohnike K, et al. Pasireotide treatment for severe congenital hyperinsulinism due to a homozygous ABCC8 mutation. Ann Pediatr Endocrinol Metab 2021;26:278–83.




15. Ashcroft FM, Puljung MC, Vedovato N. Neonatal diabetes and the KATP channel: from mutation to therapy. Trends Endocrinol Metab 2017;28:377–87.



16. Rubio-Cabezas O, Klupa T, Malecki MT; CEED3 Consortium. Permanent neonatal diabetes mellitus: the importance of diabetes differential diagnosis in neonates and infants. Eur J Clin Invest 2011;41:323–33.


17. De Franco E, Flanagan SE, Houghton JA, Lango Allen H, Mackay DJ, Temple IK, et al. The effect of early, comprehensive genomic testing on clinical care in neonatal diabetes: an international cohort study. Lancet 2015;386:957–63.



19. Urakami T. Maturity-onset diabetes of the young (MODY): current perspectives on diagnosis and treatment. Diabetes Metab Syndr Obes 2019;12:1047–56.


20. Jang KM. Maturity-onset diabetes of the young: update and perspectives on diagnosis and treatment. Yeungnam Univ J Med 2020;37:13–21.




21. Shields BM, Hicks S, Shepherd MH, Colclough K, Hattersley AT, Ellard S. Maturity-onset diabetes of the young (MODY): how many cases are we missing? Diabetologia 2010;53:2504–8.



22. Garcia-Herrero CM, Galan M, Vincent O, Flandez B, Gargallo M, Delgado-Alvarez E, et al. Functional analysis of human glucokinase gene mutations causing MODY2: exploring the regulatory mechanisms of glucokinase activity. Diabetologia 2007;50:325–33.



23. Szopa M, Wolkow J, Matejko B, Skupien J, Klupa T, Wybranska I, et al. Prevalence of retinopathy in adult patients with GCK-MODY and HNF1A-MODY. Exp Clin Endocrinol Diabetes 2015;123:524–8.


24. Dickens LT, Letourneau LR, Sanyoura M, Greeley SA, Philipson LH, Naylor RN. Management and pregnancy outcomes of women with GCK-MODY enrolled in the US Monogenic Diabetes Registry. Acta Diabetol 2019;56:405–11.




25. Caswell RC, Snowsill T, Houghton JAL, Chakera AJ, Shepherd MH, Laver TW, et al. Noninvasive fetal genotyping by droplet digital PCR to identify maternally inherited monogenic diabetes variants. Clin Chem 2020;66:958–65.




26. Colclough K, Bellanne-Chantelot C, Saint-Martin C, Flanagan SE, Ellard S. Mutations in the genes encoding the transcription factors hepatocyte nuclear factor 1 alpha and 4 alpha in maturity-onset diabetes of the young and hyperinsulinemic hypoglycemia. Hum Mutat 2013;34:669–85.


27. Steele AM, Shields BM, Shepherd M, Ellard S, Hattersley AT, Pearson ER. Increased all-cause and cardiovascular mortality in monogenic diabetes as a result of mutations in the HNF1A gene. Diabet Med 2010;27:157–61.


28. Menzel R, Kaisaki PJ, Rjasanowski I, Heinke P, Kerner W, Menzel S. A low renal threshold for glucose in diabetic patients with a mutation in the hepatocyte nuclear factor1alpha (HNF-1alpha) gene. Diabet Med 1998;15:816–20.


29. Stride A, Ellard S, Clark P, Shakespeare L, Salzmann M, Shepherd M, et al. Beta-cell dysfunction, insulin sensitivity, and glycosuria precede diabetes in hepatocyte nuclear factor-1alpha mutation carriers. Diabetes Care 2005;28:1751–6.

30. Pearson ER, Liddell WG, Shepherd M, Corrall RJ, Hattersley AT. Sensitivity to sulphonylureas in patients with hepatocyte nuclear factor-1alpha gene mutations: evidence for pharmacogenetics in diabetes. Diabet Med 2000;17:543–5.


31. Pearson ER, Boj SF, Steele AM, Barrett T, Stals K, Shield JP, et al. Macrosomia and hyperinsulinaemic hypoglycaemia in patients with heterozygous mutations in the HNF4A gene. PLoS Med 2007;4:e118.



32. Fantasia KL, Steenkamp DW. Optimal glycemic control in a patient with HNF1A MODY with GLP-1 RA monotherapy: implications for future therapy. J Endocr Soc 2019;3:2286–9.




33. Christensen AS, Haedersdal S, Stoy J, Storgaard H, Kampmann U, Forman JL, et al. Efficacy and safety of glimepiride with or without linagliptin treatment in patients with HNF1A diabetes (maturity-onset diabetes of the young type 3): a randomized, double-blinded, placebo-controlled, crossover trial (GLIMLINA). Diabetes Care 2020;43:2025–33.



34. Clissold RL, Shaw-Smith C, Turnpenny P, Bunce B, Bockenhauer D, Kerecuk L, et al. Chromosome 17q12 microdeletions but not intragenic HNF1B mutations link developmental kidney disease and psychiatric disorder. Kidney Int 2016;90:203–11.



35. Dubois-Laforgue D, Cornu E, Saint-Martin C, Coste J, Bellannr-Chantelot C, Timsit J, et al. Diabetes, associated clinical spectrum, long-term prognosis, and genotype/ phenotype correlations in 201 adult patients with hepatocyte nuclear factor 1B (HNF1B) molecular defects. Diabetes Care 2017;40:1436–43.



37. Bansal V, Boehm BO, Darvasi A. Identification of a missense variant in the WFS1 gene that causes a mild form of Wolfram syndrome and is associated with risk for type 2 diabetes in Ashkenazi Jewish individuals. Diabetologia 2018;61:2180–8.



38. Murphy R, Turnbull DM, Walker M, Hattersley AT. Clinical features, diagnosis and management of maternally inherited diabetes and deafness (MIDD) associated with the 3243A>G mitochondrial point mutation. Diabet Med 2008;25:383–99.


39. Uimonen S, Moilanen JS, Sorri M, Hassinen IE, Majamaa K. Hearing impairment in patients with 3243A→G mtDNA mutation: phenotype and rate of progression. Hum Genet 2001;108:284–9.



40. Semple RK, Sleigh A, Murgatroyd PR, Adams CA, Bluck L, Jackson S, et al. Postreceptor insulin resistance contributes to human dyslipidemia and hepatic steatosis. J Clin Invest 2009;119:315–22.



41. Nolis T. Exploring the pathophysiology behind the more common genetic and acquired lipodystrophies. J Hum Genet 2014;59:16–23.



42. Chakera AJ, Steele AM, Gloyn AL, Shepherd MH, Shields B, Ellard S, et al. Recognition and management of individuals with hyperglycemia because of a heterozygous glucokinase mutation. Diabetes Care 2015;38:1383–92.



43. Hattersley AT, Greeley SA, Polak M, Rubio-Cabezas O, Njolstad PR, Mlynarski W, et al. ISPAD Clinical Practice Consensus Guidelines 2018: the diagnosis and management of monogenic diabetes in children and adolescents. Pediatr Diabetes 2018;19 Suppl 27:47–63.



44. Nieto T, Castillo B, Nieto J, Redondo MJ. Demographic and diagnostic markers in new onset pediatric type 1 and type 2 diabetes: differences and overlaps. Ann Pediatr Endocrinol Metab 2022;27:121–5.




45. Kakleas K, Kossyva L, Korona A, Kafassi N, Karanasios S, Karavanaki K. Predictors of associated and multiple autoimmunity in children and adolescents with type 1 diabetes mellitus. Ann Pediatr Endocrinol Metab 2022;27:192–200.




46. Shields BM, McDonald TJ, Ellard S, Campbell MJ, Hyde C, Hattersley AT. The development and validation of a clinical prediction model to determine the probability of MODY in patients with young-onset diabetes. Diabetologia 2012;55:1265–72.



47. Carmody D, Park SY, Ye H, Perrone ME, Alkorta-Aranburu G, Highland HM, et al. Continued lessons from the INS gene: an intronic mutation causing diabetes through a novel mechanism. J Med Genet 2015;52:612–6.



48. Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 2015;17:405–24.




49. Abou Tayoun AN, Pesaran T, DiStefano MT, Oza A, Rehm HL, Biesecker LG, et al. Recommendations for interpreting the loss of function PVS1 ACMG/AMP variant criterion. Hum Mutat 2018;39:1517–24.




50. Brnich SE, Abou Tayoun AN, Couch FJ, Cutting GR, Greenblatt MS, Heinen CD, et al. Recommendations for application of the functional evidence PS3/BS3 criterion using the ACMG/AMP sequence variant interpretation framework. Genome Med 2019;12:3.



51. Reiner AP, Barber MJ, Guan Y, Ridker PM, Lange LA, Chasman DI, et al. Polymorphisms of the HNF1A gene encoding hepatocyte nuclear factor-1 alpha are associated with C-reactive protein. Am J Hum Genet 2008;82:1193–201.


52. Voleti B, Agrawal A. Regulation of basal and induced expression of C-reactive protein through an overlapping element for OCT-1 and NF-kappaB on the proximal promoter. J Immunol 2005;175:3386–90.

53. Owen KR, Thanabalasingham G, James TJ, Karpe F, Farmer AJ, McCarthy MI, et al. Assessment of high-sensitivity C-reactive protein levels as diagnostic discriminator of maturity-onset diabetes of the young due to HNF1A mutations. Diabetes Care 2010;33:1919–24.




54. McDonald TJ, Shields BM, Lawry J, Owen KR, Gloyn AL, Ellard S, et al. High-sensitivity CRP discriminates HNF1AMODY from other subtypes of diabetes. Diabetes Care 2011;34:1860–2.




55. Rama Chandran S, Bhalshankar J, Farhad Vasanwala R, Zhao Y, Owen KR, Su-Lyn Gardner D. Traditional clinical criteria outperform high-sensitivity C-reactive protein for the screening of hepatic nuclear factor 1 alpha maturity-onset diabetes of the young among young Asians with diabetes. Ther Adv Endocrinol Metab 2018;9:271–82.




56. Lauc G, Essafi A, Huffman JE, Hayward C, Knezevic A, Kattla JJ, et al. Genomics meets glycomics-the first GWAS study of human N-Glycome identifies HNF1α as a master regulator of plasma protein fucosylation. PLoS Genet 2010;6:e1001256.



57. Richter S, Shih DQ, Pearson ER, Wolfrum C, Fajans SS, Hattersley AT, et al. Regulation of apolipoprotein M gene expression by MODY3 gene hepatocyte nuclear factor-1alpha: haploinsufficiency is associated with reduced serum apolipoprotein M levels. Diabetes 2003;52:2989–95.

58. Bonner C, Nyhan KC, Bacon S, Kyithar MP, Schmid J, Concannon CG, et al. Identification of circulating microRNAs in HNF1A-MODY carriers. Diabetologia 2013;56:1743–51.



59. Agbu P, Carthew RW. MicroRNA-mediated regulation of glucose and lipid metabolism. Nat Rev Mol Cell Biol 2021;22:425–38.




60. Hwang JS. MODY syndrome. J Korean Soc Pediatr Endocrinol 2010;15:1–6.
61. Hattersley AT, Ashcroft FM. Activating mutations in Kir6.2 and neonatal diabetes: new clinical syndromes, new scientific insights, and new therapy. Diabetes 2005;54:2503–13.

62. Laver TW, Colclough K, Shepherd M, Patel K, Houghton JA, Dusatkova P, et al. The common p.R114W HNF4A mutation causes a distinct clinical subtype of monogenic diabetes. Diabetes 2016;65:3212–7.




63. Lee HJ, Ahn CW, Kim SJ, Song YD, Lim SK, Kim KR, et al. Mutation in hepatocyte nuclear factor-1alpha is not a common cause of MODY and early-onset type 2 diabetes in Korea. Acta Diabetol 2001;38:123–7.

64. Kim KA, Kang K, Chi YI, Chang I, Lee MK, Kim KW, et al. Identification and functional characterization of a novel mutation of hepatocyte nuclear factor-1alpha gene in a Korean family with MODY3. Diabetologia 2003;46:721–7.



65. Hwang JS, Shin CH, Yang SW, Jung SY, Huh N. Genetic and clinical characteristics of Korean maturity-onset diabetes of the young (MODY) patients. Diabetes Res Clin Pract 2006;74:75–81.


66. Shim YJ, Kim JE, Hwang SK, Choi BS, Choi BH, Cho EM, et al. Identification of candidate gene variants in Korean MODY families by whole-exome sequencing. Horm Res Paediatr 2015;83:242–51.



67. Kwak SH, Jung CH, Ahn CH, Park J, Chae J, Jung HS, et al. Clinical whole exome sequencing in early onset diabetes patients. Diabetes Res Clin Pract 2016;122:71–7.


68. Cho EH, Min JW, Choi SS, Choi HS, Kim SW. Identification of maturity-onset diabetes of the young caused by glucokinase mutations detected using whole-exome sequencing. Endocrinol Metab (Seoul) 2017;32:296–301.




69. Park SS, Jang SS, Ahn CH, Kim JH, Jung HS, Cho YM, et al. Identifying pathogenic variants of monogenic diabetes using targeted panel sequencing in an east asian population. J Clin Endocrinol Metab 2019;104:4188–98.



70. Yorifuji T, Higuchi S, Kawakita R, Hosokawa Y, Aoyama T, Murakami A, et al. Genetic basis of early-onset, maturity-onset diabetes of the young-like diabetes in Japan and features of patients without mutations in the major MODY genes: dominance of maternal inheritance. Pediatr Diabetes 2018;19:1164–72.










